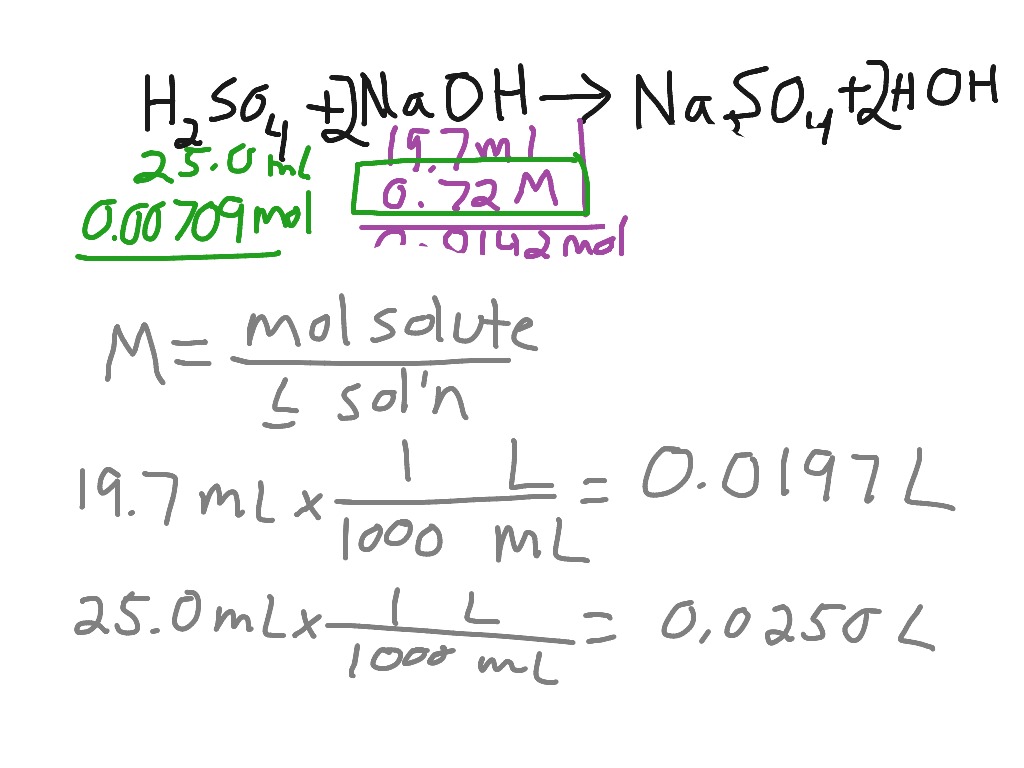
Acids and bases have been known by their properties since the early days of experimental chemistry. The word “acid” comes from the Latin acidus , meaning “sour” or “tart,” since water solutions of acids have a sour or tart taste.
Although copper doesn’t readily react with dilute hydrochloric acid and dilute sulfuric acid (low in reactivity series), if heated with nasty oily concentrated sulfuric acid you make nasty pungent irritating sulphur dioxide gas and white anhydrous copper(II) sulfate, but this is NOT a reaction on which to base its place in the metal reactivity

Sulfuric acid properties, uses, and production using the Contact Process tutorial for chemistry students.


Structure, properties, spectra, suppliers and links for: Sulfuric acid, 7664-93-9, H2SO4.


The Reactivity of Metals with Dilute Acid. The Reactivity Series. The Reaction of Metals with Dilute Acid.. Potassium, sodium, lithium and calcium all react violently with dilute sulfuric acid and dilute hydrochloric acid.
It’s important to add sulfuric acid to water and not water to concentrated sulfuric acid. Here’s why you don’t want to make a mistake.
Sulfuric acid – contact process: Sulfuric acid can be produced by reacting sulfur trioxide directly with water according to the equation below. SO 3(g) + H 2 O (l) =>H 2 SO 4(aq) /\ H = -130kJ mol-
In this experiment an insoluble metal oxide is reacted with a dilute acid to form a soluble salt. Copper(II) oxide, a black solid, and colourless dilute sulfuric acid react to produce copper(II) sulfate, giving a characteristic blue colour to the solution.


Get an answer for ‘Write and balance the equation for the reaction of hydrochloric acid (H2SO4) and sodium hydroxide to produce sodium sulfate and water.’ and find homework help for other Science questions at eNotes

general remarks. Determination of sulfuric acid concentration is very similar to titration of hydrochloric acid, although there are two important diferences.
